 Argon Medical Devices manufactures the Option™ ELITE IVC Filter. Unfortunately, it has been linked to life-threatening side effects that may require hospitalization or surgery.
Argon Medical Devices manufactures the Option™ ELITE IVC Filter. Unfortunately, it has been linked to life-threatening side effects that may require hospitalization or surgery.
Need an IVC Filter Lawyer in Texas? Collen A. Clark is a true advocate for his clients and is passionate about helping Texans. If you or a loved one has an IVC filter implant, even if you were not injured by side effects, you should contact our lawyers immediately. You may be entitled to compensation by filing a lawsuit.
Overview
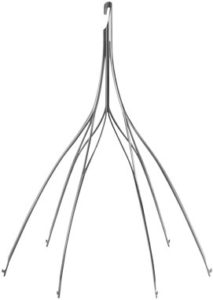
The Option™ Elite is a retrievable inferior vena cava (IVC) filter that is designed and manufactured by Rex Medical and Argon Medical Devices Inc. In January 2014, the FDA approved a new over-the-wire delivery technique that improves safety and allows for better centering of the filter within the vena cava.
Option IVC Filter Lawsuits Filed in Texas
Argon Medical Devices is facing a growing number of lawsuits from people who were injured by the Option ELITE. One lawsuit (PDF) was filed in Dallas County Circuit Court in August 2016, by a man who has the filter permanently embedded in his vena cava because it cannot be removed safely.
In October 2016, another lawsuit (PDF) was filed by a woman with an Option IVC Filter permanently embedded in her body. Doctors discovered that the filter tilted and punctured through her vena cava, where it cannot be removed without causing more serious health problems. As a result, she suffers from blood clots, chronic pain, and circulation problems. According to the lawsuit
“As long as the Option filter remains embedded in her vena cava, Plaintiff is at risk for further thrombosis and future filter fractures, migrations and perforations and tilting. She faces numerous health risks, including the risk of death.”
1st Clinical Trial Underway to Study Safety Risks
In December 2015, researchers began enrolling patients in the PRESERVE clinical trial, which will evaluate the safety of many types of retrievable filters. The study will include thousands of patients, including at least 300 with the Option™ Elite.
FDA Safety Warnings & Recommendation
In August 2010, the FDA warned that retrievable IVC filters had been linked to nearly 1,000 adverse events since 2005, including filters that fractured, migrated, or perforated the vena cava. In many cases, the patients were injured after a retrievable filter was left implanted for years — long after they were no longer at risk of a pulmonary embolism. In May 2014, the FDA recommended removing IVC filters between 29 and 54 days after implantation, so long as a patient was no longer at risk of a pulmonary embolism.
Vena Cava Filter Safety Risks
- Deep Vein Thrombosis (DVT)
- Fracture
- Embolization of broken pieces
- Vena cava perforation
- Migration to the heart or lungs
- Bleeding or hematoma
- Chronic pain
- Pulmonary embolism
- Irregular heart rhythm
- Kidney failure
- Stroke, cerebrovascular accident, or neurologic event
- Death
Complications
The Option ELITE is made of nitinol (nickel-titanium alloy) and patients who are allergic to metal could have sensitivity reactions. When serious complications occur, patients may need surgical repair or intervention. Internal bleeding may require transfusions or intravenous fluid and medications. Infections may also require antibiotics and surgical drainage. In some cases, broken filters can’t be safely removed from a patient.
IVC Filter Lawsuit Information
Free Confidential Lawsuit Evaluation: If you or a loved one has an IVC Filter, our lawyers would like to speak with you immediately. We are evaluating every case regardless of whether you experienced side effects or not. You may be entitled to compensation by filing a lawsuit. Please use the form below to contact our lawyers or call us toll free 24 hours a day at (866) 879-3040.
Need an Option IVC Filter Lawyer in Texas?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas Option ELITE IVC filter lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: