 The longer the Crux vena cava filter remains implanted in a patient, the higher the risk of complications like fracture and movement. The manufacturer is involved in a massive clinical trial to fully investigate these risks.
The longer the Crux vena cava filter remains implanted in a patient, the higher the risk of complications like fracture and movement. The manufacturer is involved in a massive clinical trial to fully investigate these risks.
Need an IVC Filter Lawyer in Texas? Collen A. Clark is a true advocate for his clients and is passionate about helping Texans. If you or a loved one has an IVC filter implant, even if you were not injured by side effects, you should contact our lawyers immediately. You may be entitled to compensation by filing a lawsuit.
What is the Volcano Crux Vena Cava Filter?
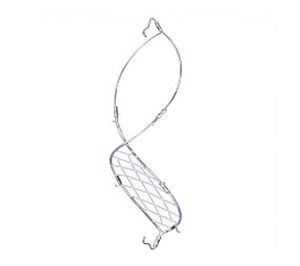
Volcano Corporation manufactures the Crux Vena Cava Filter, a medical device that is implanted in the inferior vena cava (IVC) to catch blood clots before they travel to the lungs and cause a life-threatening pulmonary embolism.
The Crux filter has a unique double helical design, which is intended to facilitate bidirectional retrieval (either through the femoral or jugular veins) and prevent tilting, migration, and penetration through the wall of the vena cava.
PRESERVE Study Investigates Crux Filter Safety
The PRESERVE clinical trial is the first large-scale, real-world study to investigate the safety of IVC filters, including the Crux filter and six other filters on the market.
The 5-year study began in early 2015 and will enroll about 2,100 patients, including at least 300 patients implanted with the Crux filter. It will specifically address concerns that retrievable filters can move or break, potentially causing significant health problems for patients.
What is the problem?
The FDA recommends removing IVC filters as soon as the patient no longer needs protection from blood clots — ideally, between 29 and 54 days after implantation.
However, studies indicate that only about 30-50% of retrievable filters are ever removed. The longer a filter remains implanted, the higher the risk of complications. Another problem is that retrievable filters were only studied for short-term safety and the long-term risks are unknown.
Safety Risks of IVC Filters
In August 2010, the FDA issued a Safety Communication after receiving 921 adverse event reports involving IVC filters since 2005. The agency was concerned about severe side effects, such as:
- Filter fracture
- Perforation of filter legs through the wall of the vena cava
- Internal bleeding (hemorrhage or hematoma)
- Organ perforation (heart, lungs, etc.)
- Movement of the filter or broken pieces to other parts of the body (embolization)
- Filter is clogged with blood clots and blocks blood circulation
- Surgery may not be able to remove broken pieces
- And more
Crux Vena Cava Filter Lawsuit
Hundreds of people who were injured by vena cava filters have already filed lawsuits, but not as part of a class action lawsuit. Instead, they are individual lawsuits that have been centralized in federal court.
Most of the cases are coordinated in a “mass-tort” or Multi-District Litigation (MDL) against Cook Medical and C.R. Bard. These manufacturers are accused of selling defective and dangerous medical devices without warning about potential risks.
IVC Filter Lawsuit Information
Free Confidential Lawsuit Evaluation: If you or a loved one has an IVC Filter, our lawyers would like to speak with you immediately. We are evaluating every case regardless of whether you experienced side effects or not. You may be entitled to compensation by filing a lawsuit. Please use the form below to contact our lawyers or call us toll free 24 hours a day at (866) 879-3040.
Need a Crux IVC Filter Lawyer in Texas?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas Crux IVC filter lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: