 The Cordis OptEase and TrapEase IVC filters are designed to catch blood clots. Unfortunately, studies have found high rates of filter fracture, especially when they are not removed within a few months.
The Cordis OptEase and TrapEase IVC filters are designed to catch blood clots. Unfortunately, studies have found high rates of filter fracture, especially when they are not removed within a few months.
Need an IVC Filter Lawyer in Texas? Collen A. Clark is a true advocate for his clients and is passionate about helping Texans. If you or a loved one has an IVC filter implant, even if you were not injured by side effects, you should contact our lawyers immediately. You may be entitled to compensation by filing a lawsuit.
UPDATE: Lawyers Seek “Mass-Tort” for Cordis IVC Filter Lawsuits
June 16, 2016 — Lawyers want to transfer Cordis IVC filter lawsuits involving 115 personal injury plaintiffs into one federal court, arguing the cases are a “mass-tort” that should be consolidated. Click here to read more.
Cordis IVC Filters
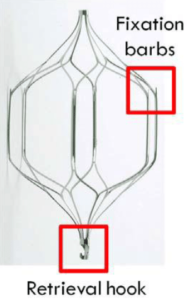
Cordis Corporation manufactures the OptEase and TrapEase IVC filters. These retrievable wire implants are designed to filter blood clots in patients who can’t take blood-thinning medications. They are implanted in the inferior vena cava (IVC) to prevent pulmonary embolisms.
What is the problem?
OptEase and TrapEase are temporary IVC filters that must be removed as soon as the patient’s risk of blood clots subsides — ideally within 29 and 54 days after implantation, according to the FDA.
The problem is that use of retrievable filters has skyrocketed, but most of them are never removed.
The longer an IVC filter remains implanted, the higher the risk of complications. The FDA has received thousands of adverse event reports and issued a Safety Communication about health risks, such as:
- Fracture
- Embolization of fragments to the heart or lungs
- Migration
- Tilt
- Organ damage
- Internal bleeding
- Deep Vein Thrombosis (DVT)
- Circulation problems
- Unable to retrieve filter or broken pieces
Study Finds 50% Fracture Risk from Cordis IVC Filter
In November 2011, Japanese researchers reported a 50% fracture-rate from the Cordis TrapEase IVC filter after 50 months. The study involved 20 patients and no life-threatening events occurred. Of the 10 fractured filters, eight had a single fracture and two had multiple fractures.
Cordis OptEase IVC Filter Recall
In March 2013, Cordis recalled OptEase IVC filters because they could be implanted backwards. The fixation barbs would point in the wrong direction and there would be nothing to stop the filter from moving into the patient’s heart. The recall includes about 33,000 filters sold from 2010-2013.
OptEase Approved Without Safety Studies
The OptEase has a unique balloon shape. Other IVC filters are cone-shaped — including the Recovery, which was withdrawn from the market in 2005, and the Günther Tulip. When the FDA approved OptEase in 2003, these cone-shaped devices were cited as “equivalent” in the 510(k) application, which allowed the OptEase on the market without requiring new safety studies.
IVC Filter Lawsuits
Approximately 1,000 lawsuits have been filed against manufacturers of IVC filters. These lawsuits are not part of a class action. Instead, they are individual lawsuits that have been centralized in federal court. As of May 2016, C.R. Bard was facing about 550 IVC filter lawsuits in Arizona (MDL No. 2641) and Cook Medical was facing nearly 400 lawsuits in Illinois (MDL No. 2570).
IVC Filter Lawsuit Information
Free Confidential Lawsuit Evaluation: If you or a loved one has an IVC Filter, our lawyers would like to speak with you immediately. We are evaluating every case regardless of whether you experienced side effects or not. You may be entitled to compensation by filing a lawsuit. Please use the form below to contact our lawyers or call us toll free 24 hours a day at (866) 879-3040.
Need a Cordis IVC Filter Lawyer in Texas?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas Cordis IVC filter lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: