 The FDA is recalling anesthesia breathing circuits manufactured by Teleflex Medical because they can crack. This could lead to serious side effects, including delayed treatment, breathing problems, or even death.
The FDA is recalling anesthesia breathing circuits manufactured by Teleflex Medical because they can crack. This could lead to serious side effects, including delayed treatment, breathing problems, or even death.
Need a Texas Anesthesia Injury Lawyer? Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged. If you or a loved one was injured by a defective anesthesia breathing circuit, you should contact our lawyers immediately. You may be entitled to compensation by filing a lawsuit in Texas.
Overview
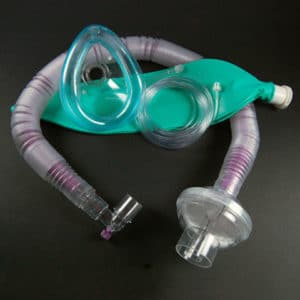
Anesthesia breathing circuits are medical devices that deliver a mixture of anesthesia gases and/or other gases from a mechanical ventilator to a patient. It provides both an inhalation and exhalation route and may include a connector, adaptor, and Y-piece.
Teleflex Recalls Hudson HCI Pediatric Anesthesia Breathing Circuit
September 8, 2014 — Teleflex Medical is warning customers not to use the Hudson RCI Pediatric Anesthesia Breathing Circuit because the ends of the circuit can crack of break before or during use.
This could cause serious health consequences, including delayed treatment of the patient, breathing difficulties, or death if the circuit is not replaced immediately. The recalled device is used in hospitals to care for pediatric patients.
FDA Issues Class I Recall
The U.S. Food and Drug Administration (FDA) has issued a Class I recall for approximately 27,176 breathing circuits that were sold throughout the United States and worldwide.
The recall applies to breathing circuits manufactured from March 2013 to July 2014 and distributed from June 2013 to May 2014.
Recommendation
- Stop using the devices
- Remove the devices from inventory and stop distribution
- Return the Recall Acknowledgment Form included with the letter
- Notify any customers who may have received the devices through re-distribution
King Systems Universal Flex2 Breathing Circuit Recall
In February 2009, a Class II recall was issued for about 500,000 of the King Systems Universal Flex2 Breathing Circuit. The device is used to administer medical gases and/or anesthesia gases to a patient.
According to the FDA:
“The inner line may be severed or broken into two pieces, thus hindering the delivery of oxygen and anesthetics to the patient and possibly elevated patient carbon dioxide levels.”
In September 2013, the FDA issued a warning letter to King Systems Corp. after finding that the anesthesia breathing circuits were not manufactured in compliance with federal safety requirements.
This led to a Class I recall for the Universal Flex2 Breathing Circuit because a customer received a latex bag that was labeled as non-latex.
Need an Anesthesia Injury Lawyer in Texas?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas anesthesia injury lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: