February 24, 2014 — Teleflex Medical Inc. has recalled a tracheal tube following complaints that it can kink during use.The recalled tracheal tube is inserted into a patient’s windpipe (trachea) through the nose or mouth and used to maintain an open airway.
A kink in the tube could deprive a patient of adequate ventilation, which could lead to oxygen deprivation and death. The U.S. Food and Drug Administration (FDA) has classified the action as a Class 1 recall, which means there is a reasonable probability that the defect could cause severe injury or death.
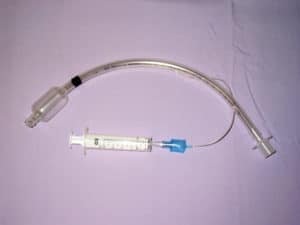
Teleflex recalled the ISIS HVT Tracheal Tube Cuffed with subglottic secretion suction port (with or without Preloaded Stylet). It has a suction port and a separate line for subglottic secretion suctioning. The device is designed to provide airway access and ventilation on a short-term or long-term basis.
The affected products were manufactured from March 2010 through December 2013. The recall was initiated on January 6 when Teleflex sent their customers an Urgent Medical Device Recall Notification.
FDA Recommendation:
“Immediately discontinue use and quarantine any products with the catalog numbers listed in the Urgent Medical Device Recall Notification. Complete the Recall Acknowledgement Form and fax it to 1-866-804-9881, Attn: Customer Service. For questions about this recall, customers can call Teleflex Medical at 1-866-804-9881, 8 am to 8 pm, Eastern Time, Monday through Friday.”
Need a Texas Medical Device Lawyer?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas medical device lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: