 October 3, 2016 — The FDA has issued another warning against using homeopathic teething gels and tablets.
October 3, 2016 — The FDA has issued another warning against using homeopathic teething gels and tablets.
The agency has received reports of seizures in infants and children who were given these products since a previous warning in 2010.
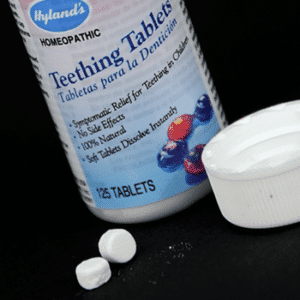
In October 2010, a recall was issued for Hyland’s Teething Tablets after the FDA received reports of belladonna toxicity in children.
Belladonna, also known as “deadly nightshade,” has a long history of use in medicine, cosmetics, and warfare. It is one of the most toxic plants in the world, but has a mild anesthetic effect in small amounts.
The FDA found inconsistent amounts of belladonna in Hyland’s Teething Tablets. The agency is currently testing more product samples and will update the public as more information is collected.
The symptoms of belladonna toxicity may include seizures, difficulty breathing, lethargy, excessive sleepiness, muscle weakness, skin flushing, constipation, difficulty urinating, or agitation.
Homeopathic teething tablets and gels are sold by CVS, Hyland’s, other retail stores, and online. The FDA is warning consumers not to use these products and throw away any in their possession.
The FDA has also issued warnings against giving infants teething gels containing anesthetics like benzocaine or lidocaine due to the risk of life-threatening side effects like methemoglobinemia.
Need a Product Liability Lawyer in Texas?
Collen A. Clark is a true advocate for his clients and is passionate about helping Texans that have been injured or wronged.
Collen’s amazing success in the courtroom and well known dedication to his clients has earned him the recognition of his peers as one of The Top Trial Lawyers in Texas.”
The Clark Firm has assembled a team of trial lawyers with more than 100 years experience, participation in over 600 jury trials and $260 million in verdicts and/or settlements. Please use the form below to contact our Texas product liability lawyers for a free lawsuit review.


 To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing:
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing: